On the other hand copper metal is less reactive than iron and zinc and hence cannot displace them from their respective salt solutions. Answer During electrolysis of aqueous copper sulphate using copper electrodes the two anions OH and SO 4 2 migrate to the anode but none of them get discharged because the copper of the anode dissolves in the solution producing copper ions and.
Solved In The Electrolysis Of Aqueous Copper Sulfate Using Chegg Com

Electrolysis Of Aqueous Cuso4 With Inert Electrodes Gives
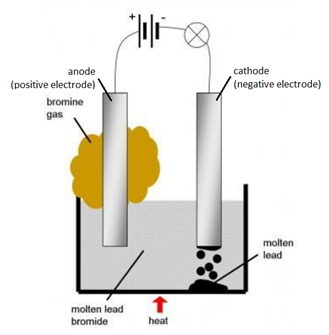
1 58 Triple Only Describe Experiments To Investigate Electrolysis Using Inert Electrodes Of Molten Compounds Including Lead Ii Bromide And Aqueous Solutions Including Sodium Chloride Dilute Sulfuric Acid And Copper Ii Sulfate And To Predict
Chapter 3 Science Class 10 MCQ Question 15.
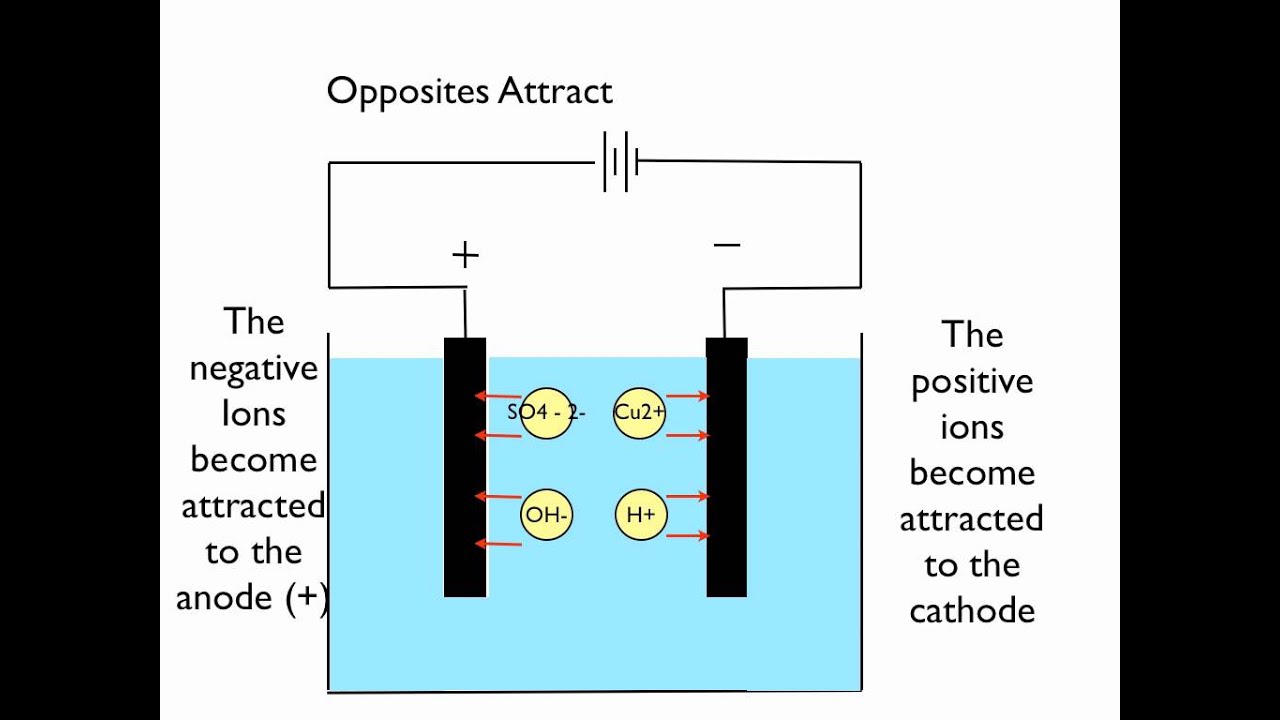
Electrolysis of copper sulphate using inert electrodes. Ion-selective electrodes 26 27 can be used to determine certain elements such as chloride. Electrolysis with inert electrodes like platinum graphite etc. - Concentrated aqueous sodium chloride using inert electrodes.
Because of iron going into solution as iron sulphate a number of holes are seen in the iron pot. We can also use electrolysis to purify substances. Cu 2 SO 4 2-H OH-At the cathode.
This instrument is so simple to use and so specific that almost any metallic element can be determined in nickel plating solutions providing suitable lamps are available. 331 Investigate the electrolysis of copper sulfate solution with inert electrodes and copper electrodes. Bare conductors or busbars used as protective conductors shall be coloured by equally broad green and yellow.
After electrolysis of a sodium chloride solution with inert electrodes for a certain period of time 600 mL of the solution was left which was found to be 1 N in NaOH. - Aqueous copperII sulphate using. There is competition between the copper ions and the hydrogen ions.
Using active electrodes results in transfer of copper metal from the anode to the cathode during electrolysis. Chemistry Notes for Students. Electrolysis of copper 11 sulphate solution using copper electrodes.
Apabila kayu uji menyala diletakkan di mulut ujian tabung uji. Iron is more reactive than copper. The copper sulphate is ionised in the aqueous solution.
The blue colour of aqueous copper sulphate fades when it is electrolysed using platinum electrodes. The circuit is completed by eg. CuSO 4 Cu 2.
Electrolysis of copper II sulphate solution using copper electrodes participating electrodes The ions present in the solution are. The electrolysis of copperII sulphate solution using a copper anode. Which inert gas electron configuration do the Cl atoms in Cl 2 molecule.
Since oxidation of water has more positive potential oxygen will be evolved at the anode. Platinum or graphite electrodes are inert and do not take part in electrolysis while metallic electrodes eg. SECONDARY STAGE CHEMISTRY BOOK ONE FOR CLASS IX.
330 Explain the formation of the products in the electrolysis of copper sulfate solution using copper electrodes and how this electrolysis can be used to purify copper. E Complete the following table for the electrolysis of 01 mol dm3 copperII sulphate solution using carbon electrodes. Na e Na at cathode Chloride ions migrate the other way toward the anode.
Instead of using carbon electrodes in a copper sulphate solution we can use copper electrodes. The ions present in the solution are. The solution becomes blue due to the formation of copper sulphate.
Calculate the percentage theoretical yield of NaOH obtained. If the two electrodes are connected using an external wire the electrons produced by the oxidation of zinc travel through the wire and enter into the copper cathode where they reduce the copper ions present in the solution and form copper atoms that. Hence when a solution of CuSo 4 is kept in an iron pot iron slowly displaces copper from the solution and blue colour of the solution keeps fading away.
Electrolysis Of Dilute Copper Sulphate Coursework what you pay for and thats what you will get 1010 times. - Dilute sulphuric acid using inert electrode. The study programs for scientific and technical degrees include classroom theoretical teaching analysis and resolution of real exercises and some more practical teaching in the laboratory.
The reason behind this displacement reaction is that copper is more reactive than silver. Copper nickel silver are active and take part in electrolysis. The external copper wiring and the usually inert electrodes like graphite form of carbon or.
The positive ions are attracted to the negative cathode. Free PDF download of Class 10 Science Chapter 3 - Metals and Non-metals Revision Notes Short Key-notes prepared by expert Science teachers from latest edition of CBSENCERT books. During electrolysis of copper sulphate solution using platinum electrode oxygen gas is liberated but on using copper electrode oxygen gas is not liberated.
The electrolysis of an aqueous solution of copper sulphate using copper electrodes ie. Electrolysis of molten lead bromide is considered to be a reaction in which oxidation and reduction go side by side ie a redox reaction. Inert electrodes do not react with the electrolyte or the products and so does not undergo any changes.
When a lighted wooden splinter is placed near the mouth of the test tube. Using electrolysis electrolysis of copper ii sulfate solution learn chemistry 3 ways to make copper sulphate in a science lab wikihow electrochemistry wikipedia thomas greenbowe department of chemistry and biochemistry varmint al s handloading reloading page oxidation of benzoin. Use of Bi-Colour CombinationGreen and YellowThe bi-colour combination green and yellow greenyellow shall be used for identifying the protective conductor and for no other purposeThis is the only colour code recognized for identifying the protective conductor.
The laboratory teaching focuses on the application of the. The reaction is CuSO 4 aq Fes FeSO 4 aq Cus. During the electrolysis of aqueous copper sulphate between copper electrodes the sulphate and hydroxyl ions remain as spectator ions.
The invention of the Atomic Absorption Spectrometer AAS radically changed the analytical position. Copper sulphate solution is ionised by the following chemical equation. In the electrolysis of brine sodium ions migrate to the cathode where electrons enter the melt and are reduced to sodium metal.
A similar change happens if you electrolyse copperII sulphate solution using copper electrodes. Electroplating is a general name for processes that produce a metal coating on a solid substrate through the reduction of cations of that metal by means of a direct electric currentThe part to be coated acts as the cathode negative electrode of an electrolytic cell. Electrolysis is the breaking down of a substance into its component elements by passing an electric current through a liquid or solution.
The process of electrolysis involves using an electric current to bring about a chemical change and make new chemicals. Copper is deposited at the cathode as you would expect but instead of oxygen being given off at the anode copperII ions go into solution. During the same period 3175 g of copper was deposited in the copper voltameter in series with the electrolytic cell.
Aluminium metal is being extracted by the electrolysis of molten aluminium oxide using carbon electrodes. The electrolyte is a solution of a salt of the metal to be coated. And the anode positive electrode is usually either a.
ÿState the ions present name the products and give the electrodes reactions in the electrolysis of - Molten sodium chloride using inert electrodes. Impure copper can be purified using electrolysis. What changes in the colour of iron nails and copper sulphate solution do you observe after keeping the iron nails dipped in copper sulphate solution for about.
The zinc ions pass into the solution. A pop sound is produced.
1

Electrolysis Of Copper Sulphate Youtube
1

Electrolysis Of Aqueous Solutions Video Lesson Transcript Study Com

Electrolysis Of Copper Sulphate Using Inert Electrodes Youtube

Core Practical Electrolysis Of Copper Ii Sulfate Edexcel Gcse Chemistry Revision Notes

Electrolysis Copper Sulfate Solution With Copper Carbon Graphite Electrodes Electroplating Half Equations Products Anode Cathode Apparatus Electrolyte Cell Gcse Chemistry Ks4 Science Igcse O Level Revision Notes
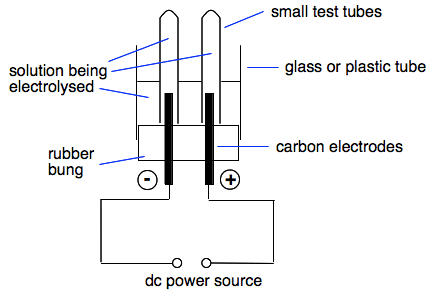
Electrolysis Of Solutions With Inert Electrodes


